
Using the table for standard enthalpy of formation, solve 2 CO (g) + O2 (g) --> 2 CO2 (g) - Brainly.com
![SOLVED: Table 2C.4* Standard enthalpies of formation (4,He) and combustion (4He) of organic compounds at 298 K 4,He /(kJ mol-!) +49.0 4 He/(k] mol-1) Benzene; CHs(l) Ethane; C,Hs(g) Glucose; CbH,Os(s) Methane, CH ( SOLVED: Table 2C.4* Standard enthalpies of formation (4,He) and combustion (4He) of organic compounds at 298 K 4,He /(kJ mol-!) +49.0 4 He/(k] mol-1) Benzene; CHs(l) Ethane; C,Hs(g) Glucose; CbH,Os(s) Methane, CH (](https://cdn.numerade.com/ask_images/34cc09f6e28a4eb6b0ebc24db7161382.jpg)
SOLVED: Table 2C.4* Standard enthalpies of formation (4,He) and combustion (4He) of organic compounds at 298 K 4,He /(kJ mol-!) +49.0 4 He/(k] mol-1) Benzene; CHs(l) Ethane; C,Hs(g) Glucose; CbH,Os(s) Methane, CH (

Table II from Large-scale calculations of gas phase thermochemistry: Enthalpy of formation, standard entropy, and heat capacity | Semantic Scholar
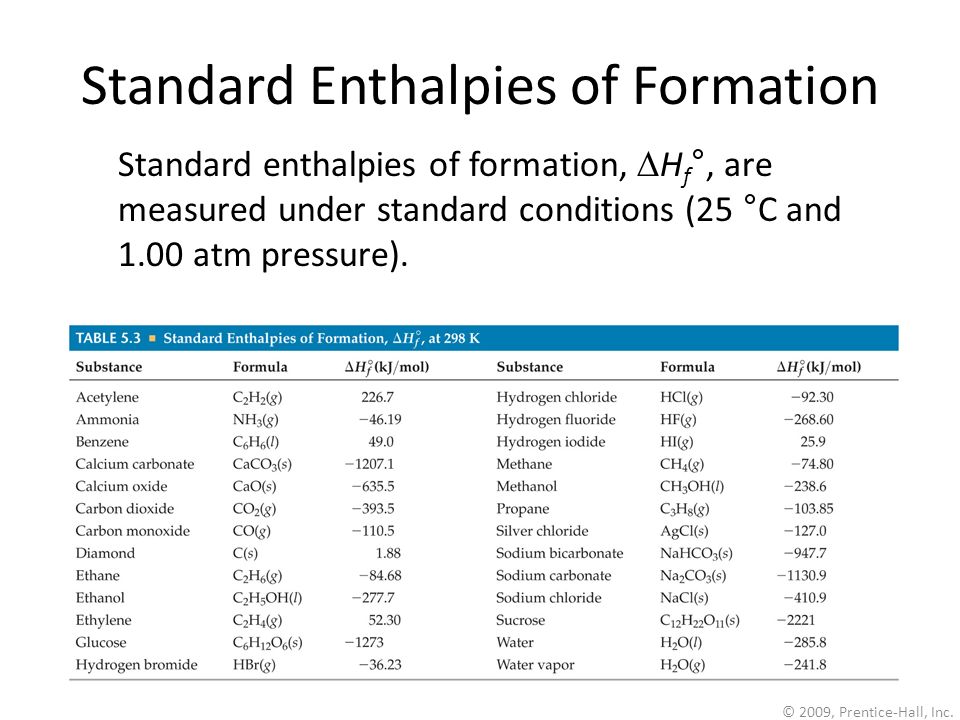
2009, Prentice-Hall, Inc. Enthalpies of Formation An enthalpy of formation, H f, is defined as the enthalpy change for the reaction in which a compound. - ppt download

Standard Enthalpies of Formation Table .pdf - Table 3.4 Standard Enthalpies and Gibbs Energies of Formation at 298.15 K 25CJT Joules per mole of the | Course Hero

Table VI from Large-scale calculations of gas phase thermochemistry: Enthalpy of formation, standard entropy, and heat capacity | Semantic Scholar
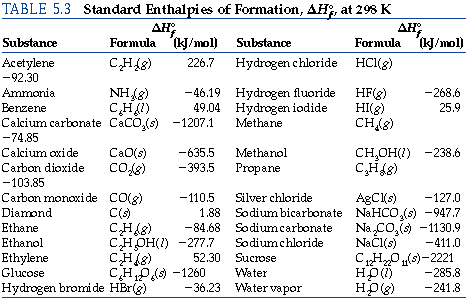
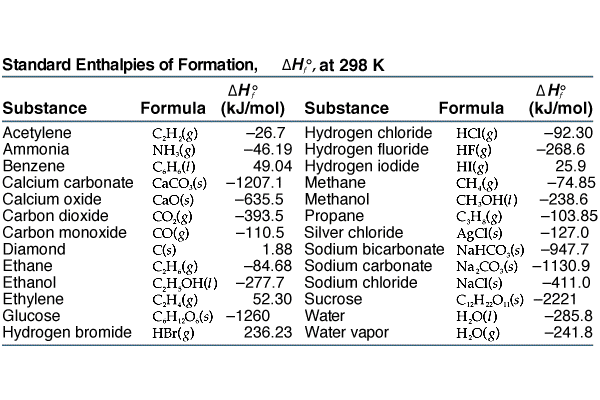
![Solved Problem 9 [ /4] Use the data table of standard | Chegg.com Solved Problem 9 [ /4] Use the data table of standard | Chegg.com](https://media.cheggcdn.com/media/066/066bdf3a-bf54-46a1-8783-f99d1f4e57cf/phpeIOgWd.png)
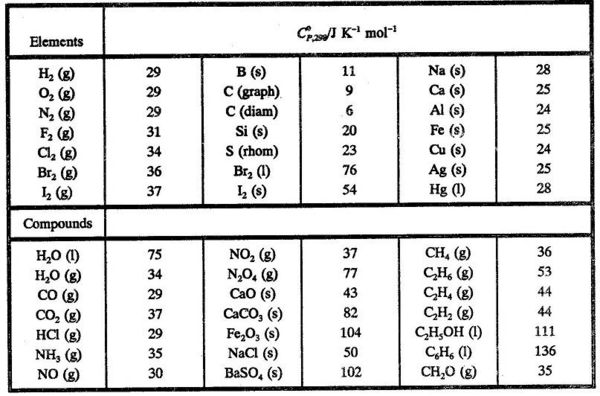

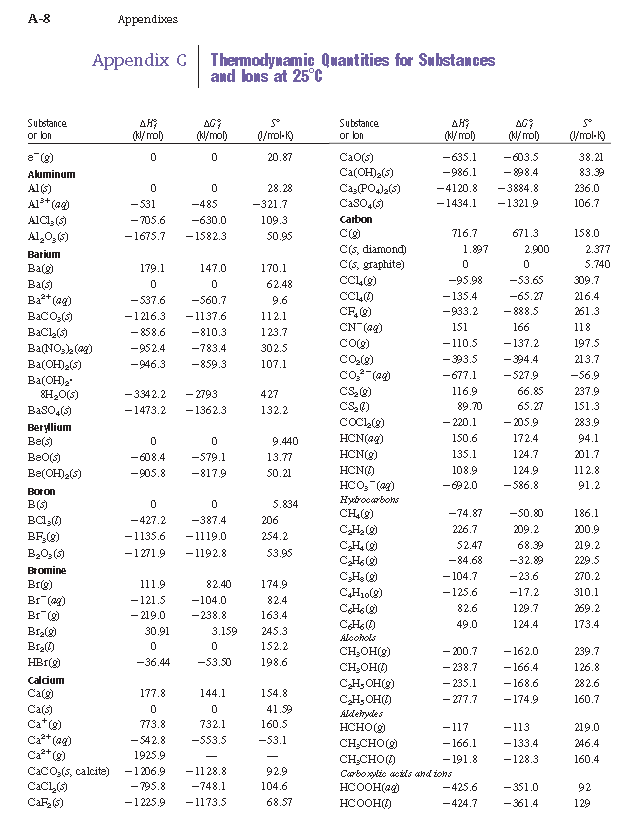
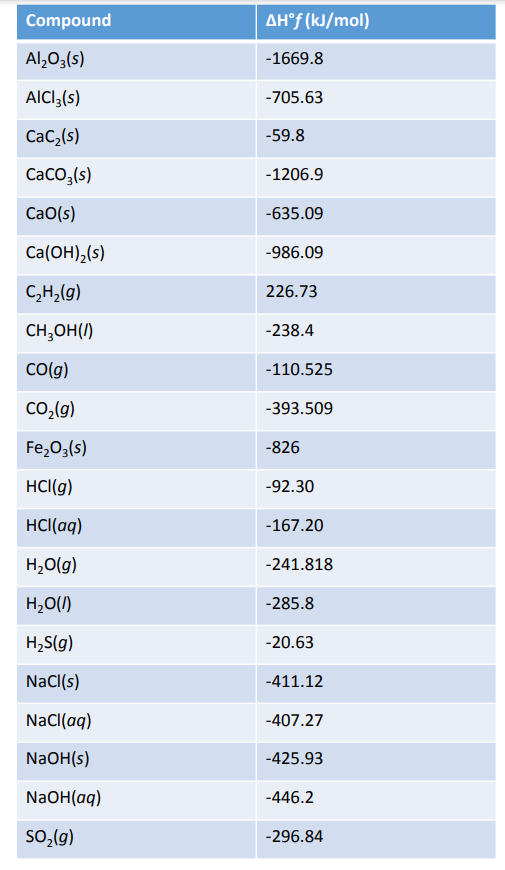


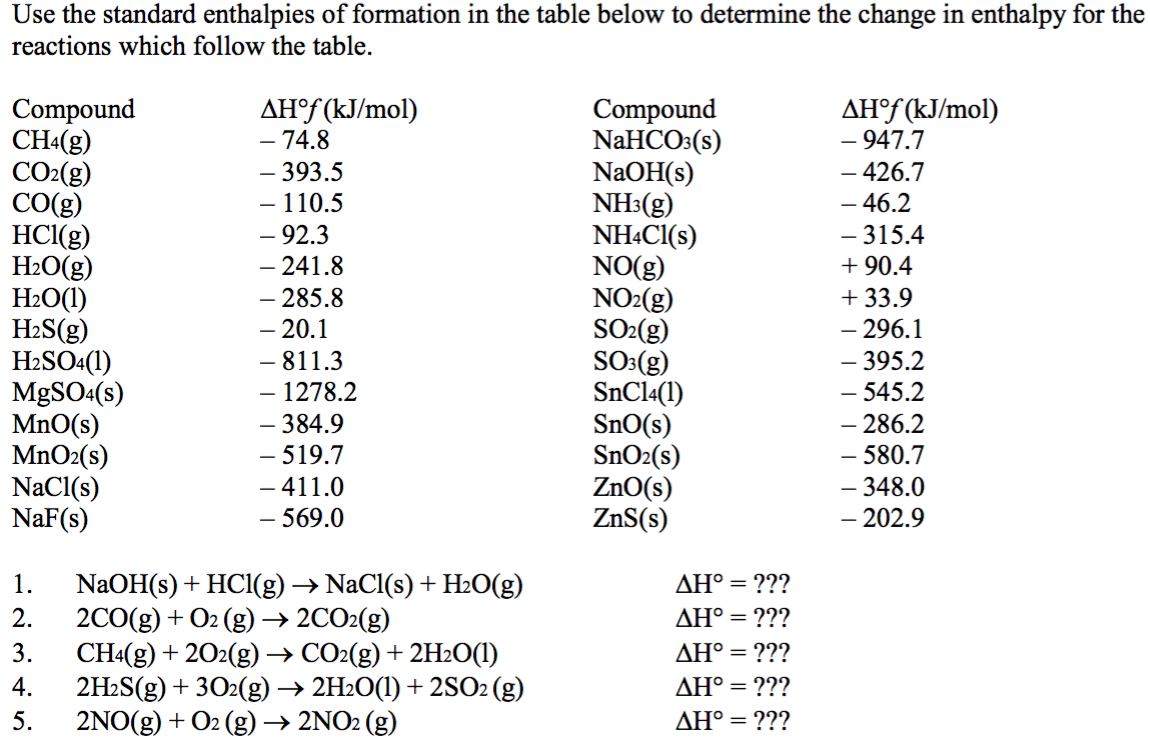
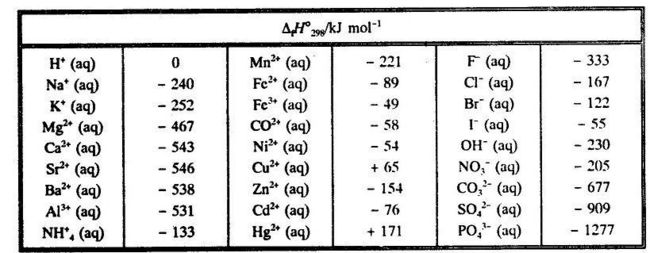
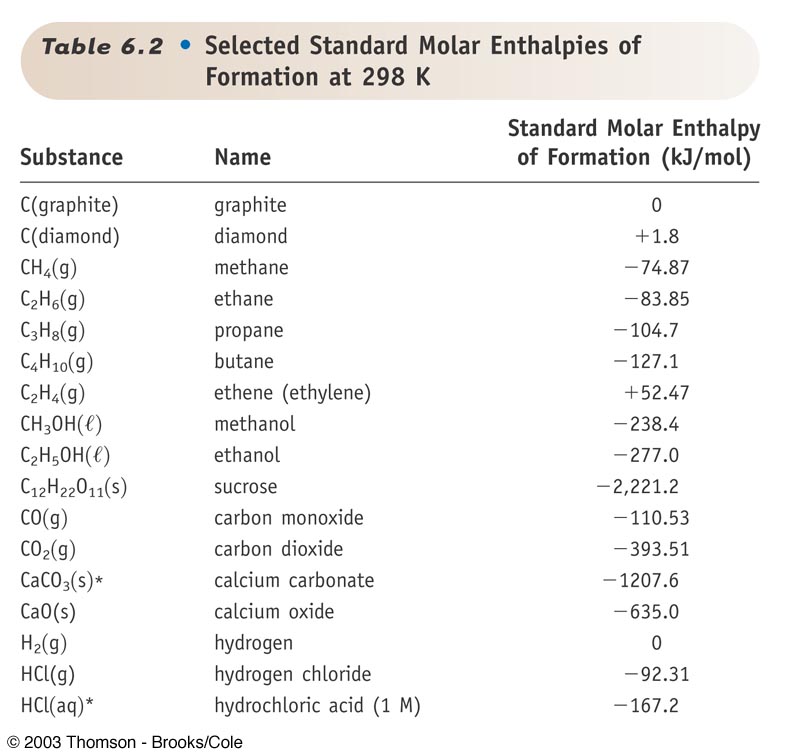



![Solved 2.[25 pts] Use the table 6.3 for the following | Chegg.com Solved 2.[25 pts] Use the table 6.3 for the following | Chegg.com](https://media.cheggcdn.com/study/f5b/f5bb4d08-1811-4eb0-ba30-8c6c0c0cdfe2/image)

