IJMS | Free Full-Text | The Fight against the Carcinogenic Epstein-Barr Virus: Gut Microbiota, Natural Medicines, and Beyond

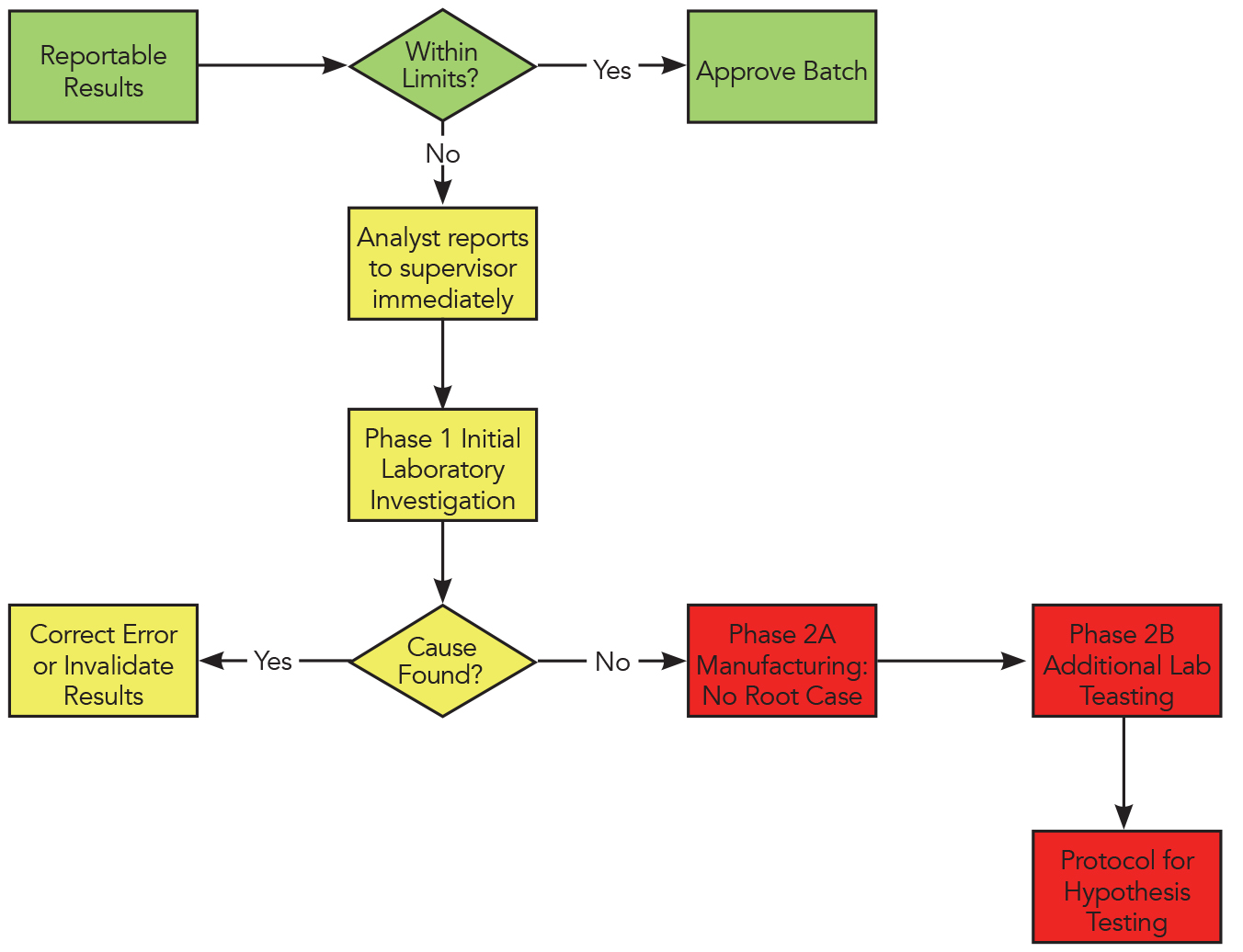
Understanding cGMPS – What Attorneys Need to Know The Nuts + Bolts of cGMPS July 10, CFR Parts 210 and 211 cGMP case law Andrew D. Bos Senior Director. - ppt download
![Laboratory Investigations and the Importance of the Barr Decision (Part 211.192) [Video] - LearnGxP: Accredited Online Life Science Training Courses Laboratory Investigations and the Importance of the Barr Decision (Part 211.192) [Video] - LearnGxP: Accredited Online Life Science Training Courses](https://learngxp.com/wp-content/uploads/2016/05/ELM-108-03-Laboratory-Investigations-and-the-Importance-of-the-Barr-Decision-Part-211.192.png)
Laboratory Investigations and the Importance of the Barr Decision (Part 211.192) [Video] - LearnGxP: Accredited Online Life Science Training Courses
Over the last twenty years, plaintiff's corporate ownership has changed. This is not an issue here. Throughout this Opinion,