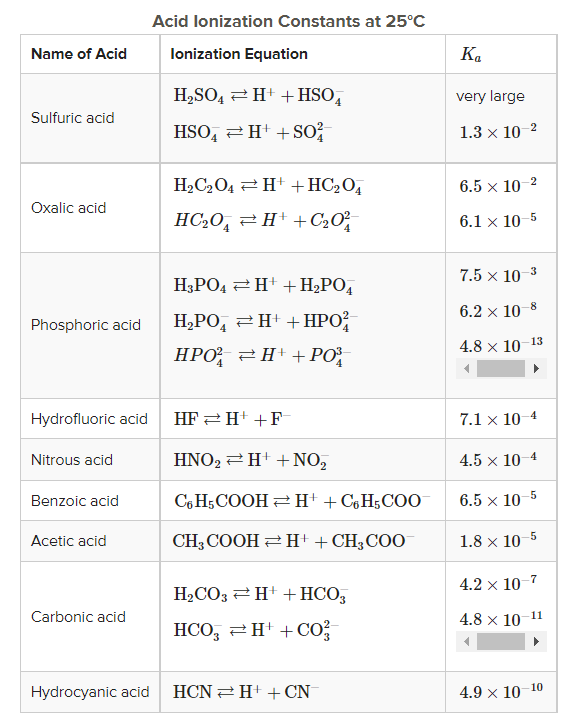
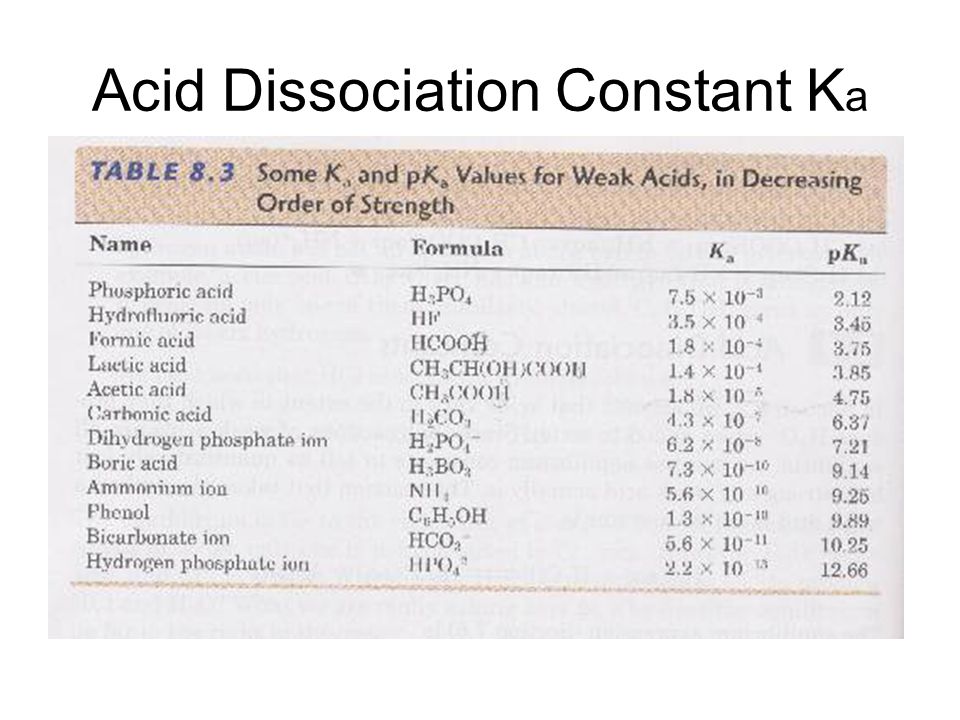
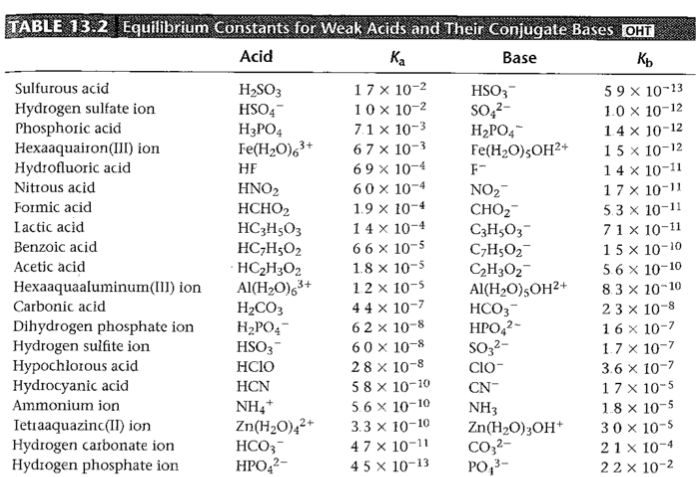
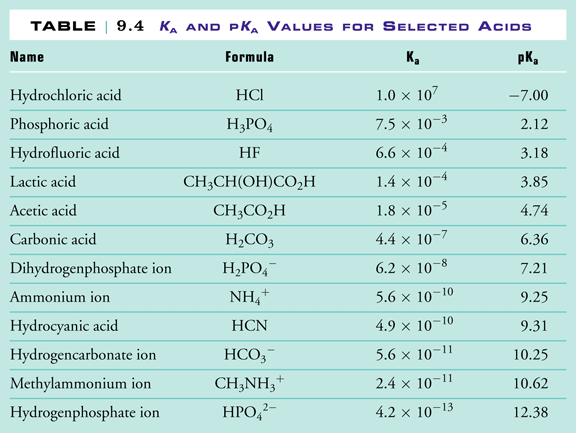
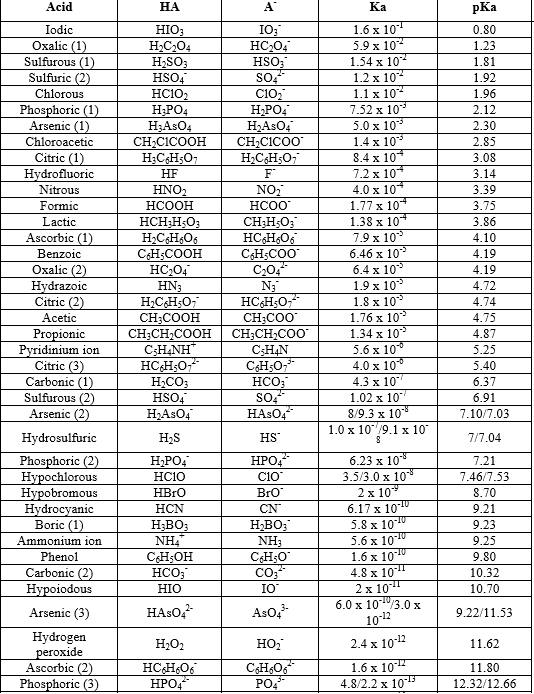
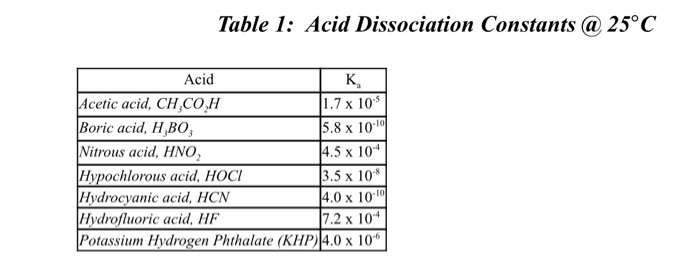
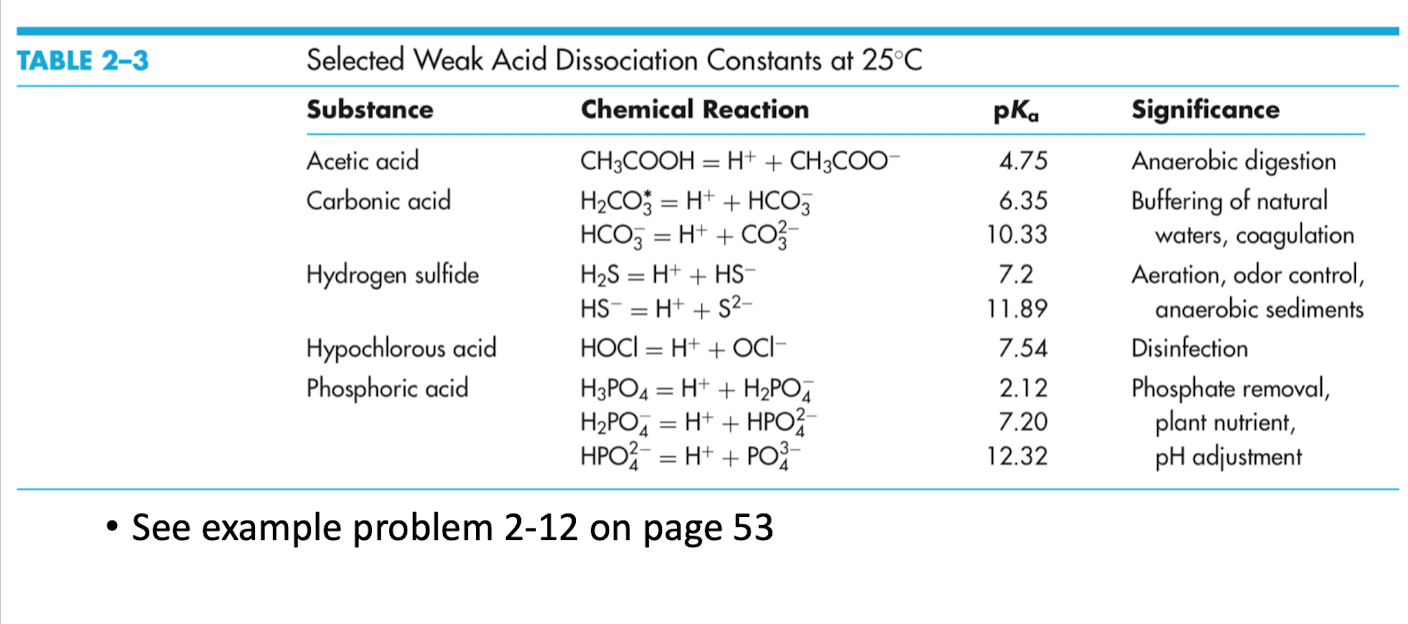
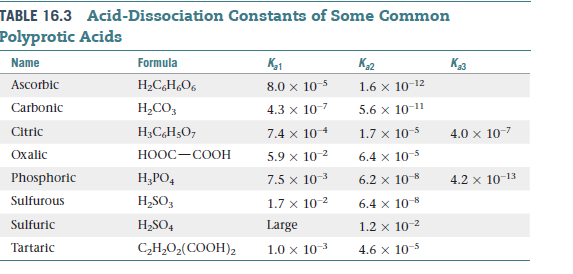
Table 1 from Association Constant of Acetic Acid in 1.02 m NaCl Aqueous Medium at Elevated Temperatures and Pressures | Semantic Scholar

Table 2 from Dissociation Constant of N-(2-Acetamido)-Iminodiacetic Acid Monosodium (ADA) from (278.15 to 328.15) K | Semantic Scholar

Table 1 from Ionization constant of 5-5'-diethylbarbituric acid from 0-degrees to 60-degrees-C | Semantic Scholar

Table 3 from Measurement of dissociation constants (pKa values) of organic compounds by multiplexed capillary electrophoresis using aqueous and cosolvent buffers. | Semantic Scholar